The Normal Aging Brain Collection Amsterdam (NABCA) is acquiring a comprehensive collection of post-mortem neuroimaging and brain tissue of the aging human brain. All data is made available to the reseach community through a Material Transfer Agreement (MTA; see application procedure). Please find below more information on how we collect imaging data and brain tissue; and which images and brain regions are available. For a publication of our pipeline, please click on this open access link to Neuroimage: Clinical.
Donor inclusion
NABCA donors become available through a collaboration with the body donation program at the department of Anatomy and Neurosciences at Amsterdam UMC – location VUmc. Anyone within the Netherlands over the age of 18 can donate their body (click for more information). A subset of these donors, which fall within the NABCA in- and exclusion criteria, will be included in our pipeline. In-/exclusion criteria involve a short time between death and arrival at Amsterdam UMC, as well as a non-neurological cause of death.
In-situ 3T MRI scanning
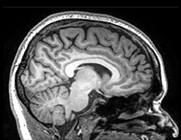
We have developed the ‘Amsterdam postmortem protocol’ in order to perform in-situ high-field structural MRI imaging using the 3 Tesla Signa-MR750 General Electric Medical System scanner at Amsterdam UMC – location VUmc within 4-10 hours after death.
Dissection

After the in situ 3T MR scanning, the autopsy is performed by a neuropathologist using a standardized protocols (special requests possible). Brain tissue samples of cortical, subcortical and brainstem regions are collected.
Ex vivo high-field 7T MRI scanning
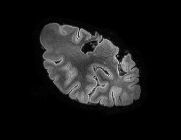
The right hemisphere and brainstem are scanned using ultra-high-field strength MR imaging (7 Tesla) at LUMC in Leiden.
Neuropathological assessment
Extensive neuropathological assessment includes evaluation of lesions in cortical, subcortical and brainstem regions using immunohistochemistry, silver staining techniques and histological staining by experienced neuropathologist.
MR images and tissue availability
NABCA can provide MRI images (3T and 7T) and brain tissue (snap frozen and FFPE) of non-neurological controls to researchers affiliated with universities, other non-profit organizations, and pharmaceutical companies in collaboration with research institutes, all over the world.
List of FFPE tissue blocks from the standardized dissection protocol
List of frozen tissue blocks from the standardized dissection protocol